PALM is a government partnership between the National Institute of Allergy and Infectious Diseases (NIAID) of the United States of America (USA) and the Ministry of Public Health of the Democratic Republic of Congo (DRC), represented by the National Institute for Biomedical Research (INRB). This collaboration emerged from the first randomized controlled trial (RCT) on the treatment of Ebola virus disease (EVD) conducted during the 2018-2020 Ebola virus disease (EVD) outbreak in North Kivu and Ituri provinces of the DRC. This study resulted in the FDA approval of two life-saving treatments. Based on the success of this study, both parties presented the need for a sustainable partnership established to continue research on infectious diseases of mutual interest.

Program Areas
PALM aims to bring together additional scientific partners who promote a collaborative approach to solving critical infectious disease problems with global impact.
The impact and reach of PALM's work is primarily dedicated to conducting clinical trials in infectious diseases and promoting studies that can help generate important scientific evidence to advance science.
As of today, PALM has grown, with nine studies in its field of activity, including interventional and observational studies, across nine sites in the DRC that are under development or ongoing.
PALM has also expanded its scope to include other infectious diseases of global concern, as COVID-19 and MPOX. PALM supports activities that contribute to the successful operation of the program, including several large biobanks containing hundreds of thousands of samples from recent Ebola, COVID-19, and MPOX outbreaks. In collaboration with the INRB, PALM’s goal is to continue to grow as a resource for infectious disease research and improving global health.
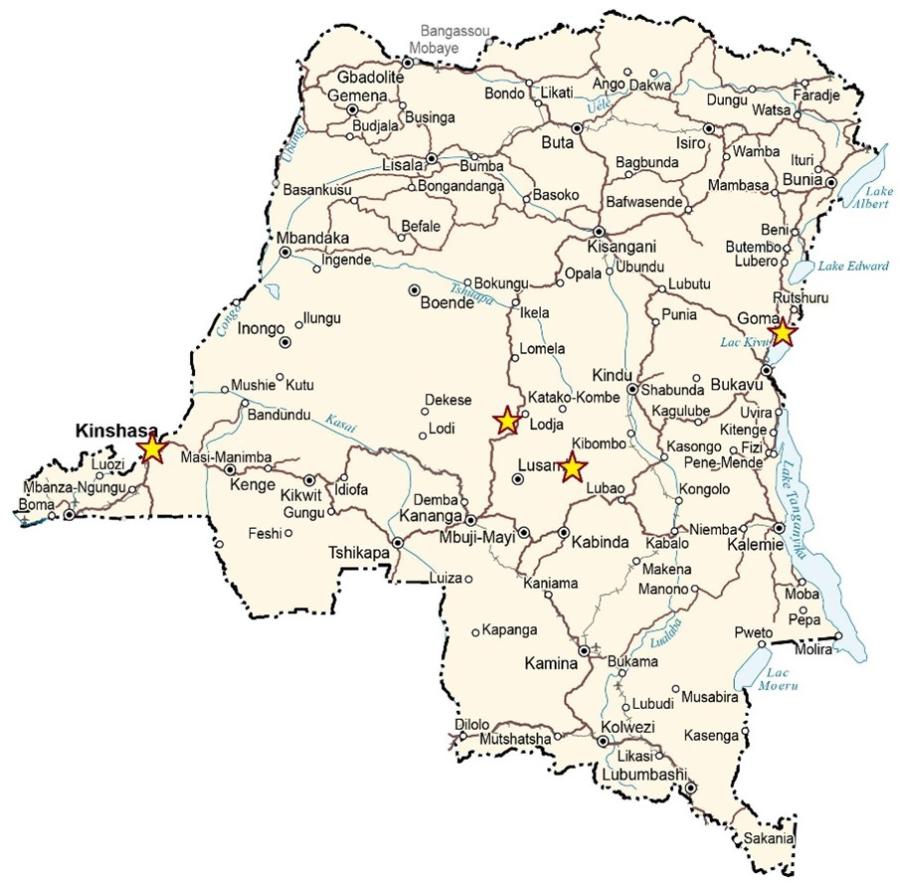
The map above shows the locations of the sites within the country (from left to right—Saint-Joseph Hospital/Kinshasa, INRB Campus/Kinshasa, Kole, Tunda, Beni, Mangina, Butembo and Katwa).
PALM Accomplishments
On October 14, 2020, the United States Food and Drug Administration (FDA) approved the first treatment for Zaire ebolavirus infection in adult and pediatric patients. Inmazeb is manufactured by Regeneron Pharmaceuticals and was one of the therapeutics used in PALM001 , where it was known as REGN-EB3. The PALM team is working on the local approval of Inmazeb in the DRC.
On December 21, 2020, the FDA approved another treatment for Zaire ebolavirus infection in adult and pediatric patients. Ebanga, manufactured by Ridgeback Biotherapeutics, LP, was another therapeutic used in PALM001, where it was known as mAb114. The PALM team is working on the local approval of Ebanga in the DRC.
The New England Journal of Medicine published A Randomized, Controlled Trial of Ebola Virus Disease Therapeutics on December 12, 2019.
The PALM RCT was honored with the Society for Clinical Trials’ prestigious David Sackett Trial of the Year Award for 2020.
On March 5, 2022, the PALM team completed enrollment of an international study on COVID-19 Vaccine to assess Immunogenicity, Reactogenicity and Efficacy. This study has generated important data on COVID vaccines immunogenicity from sub-Saharan African populations.
On October 31, 2024, the PALM team completed activities of a study on cohort events monitoring for the assessment of safety profile of MVA-BN (Jynneos) vaccine in adult personnel and research staff in the PALM 007 study in DRC.
On July 08, 2024, the PALM team completed enrollment of one of the first RCT on MPOX conducted in the DRC. The results of this trial provided important guidance for the future use of tecovirimat that did not show evidence of efficacy compared to placebo and provides data that can inform future designs and the selection of other therapeutic candidates.
PALM contributed to capacitate and promote the scientific level of local human resource. Several scientific meetings and trainings were conducted with a great impact on the operation of the program. PALM’s Operations Manager was listed by Nature Journal as one of the 10 people shaping science in 2024.
PALM’s infrastructure has grown to include biorepositories, data management center, Clinical Research Center (CRC), cold chain capabilities, pharmacies, administrative offices, and warehouse.
Clinical Studies
PALM 001 RCT (NCT03719586): 2018-2020 A Multicenter, Multi-Outbreak, Randomized, Controlled Safety and Efficacy Study of Investigational Therapeutics for the Treatment of Patients With Ebola Virus Disease that enrolled 681 participants at four sites in the DRC, Beni, Butembo, Katwa, and Mangina.
PALM 005: Assessment of immune response to EVD as modulated by vaccines and MCMs - protocol is in planning and development stage.
PALM 006/InVITE (NCT05096091): 2021-2024 multinational study of COVID-19 vaccine immunogenicity and durability that enrolled 1,100 adults at one site, St. Joseph’s Hospital, Kinshasa, DRC.
PALM 007 RCT (NCT05559099): 2022-2024 Randomized, placebo-controlled trial of the safety and efficacy of tecovirimat for the treatment of patients with monkeypox virus disease. The study enrolled 598 participants at two remote sites in the DRC, Tunda General Hospital and Kole General Hospital.
PALM 007 Extension Phase (NCT06721585): 2024-2025 open-label extension to the PALM 007 protocol for further clinical characterization of the natural history of mpox. The study is open to enrollment for up to 400 participants. The study is enrolling participants at two remote sites in the DRC, Tunda General Hospital and Kole General Hospital.
PALM 008 (NCT05734508): 2023-2024 Cohort events monitoring study for the assessment of safety profile of MVA-BN (Jynneos) vaccine in adult personnel and research staff in the PALM-007 study in DRC. The study enrolled 500 participants across three sites in the DRC, Kinshasa, Tunda, and Kole.
PALM 009: Screening EVD suspects for VHF and pathogens of interest from eastern DRC EVD outbreaks during 2018-2022. This is a non-clinical study of existing samples in the PALM biorepository that is under development.
PALM 010: Mpox vaccine RCT in DRC – protocol is in planning and development stage.
PALM 011: Mpox therapeutic RCT in DRC (other drugs) – protocol is in planning and development stage.
Publications
A Randomized, Controlled Trial of Ebola Virus Disease Therapeutics, NEJM, 2019
Ebola virus persistence and disease recrudescence in the brains of antibody-treated nonhuman primate survivors, Science Transaltional Medicine, 2022
Counting Monkeypox Lesions in Patient Photographs: Limits of Agreement of Manual Counts and Artificial Intelligence, Journal of Investigative Dermatology, 2022
Neurologic Complications of Smallpox and Monkeypox: A Review, JAMA Network, 2022
Potential complications of monkeypox, Lancet Neurol, 2022
Design of an observational multi-country cohort study to assess immunogenicity of multiple vaccine platforms (InVITE), PLos One, 2022
On the importance and challenges of global access to proven life-saving treatments for Ebolavirus, The LANCET Infectious Diseases, CORRESPONDENCE, 2023
SARS-CoV-2 seroprevalence in vaccine-naïve participants from the Democratic Republic of Congo, Guinea, Liberia, and Mali, International Journal of Infectious Disease, 2024
The surge of mpox in Africa: a call for action, The Lancet Global Health, 2024
Sustained Human Outbreak of a New MPXV Clade I Lineage in the Eastern Democratic Republic of the Congo, Nature Medicine, 2024.
Contact Information
About CCRB
Read about Collaborative Clinical Research Branch (CCRB).

