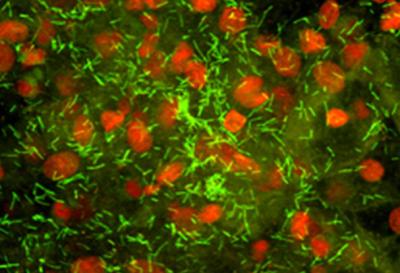
Fluorescent photomicrograph of a human vaginal biopsy from a healthy individual showing Lactobacillus (rods stained green) adhering to epithelial cells (nuclei stained red).
There are many types of bacteria in and on the human body: some cause illness while others have positive effects like aiding our digestion or producing anti-inflammatory compounds. In the vagina, healthy bacteria are thought to provide a protective barrier against unwelcome pathogens, and researchers are currently working on defining the other roles these bacteria might play.
Although there is still much to be learned about healthy vaginal flora, we do know that an infection called bacterial vaginosis (BV) can occur when the normal balance of bacteria in the vagina is disrupted. Many women with BV do not have any symptoms, but some women experience pain, itching, odor, or burning of the vagina. Once diagnosed through laboratory tests, BV can be treated with antibiotics. Unfortunately, BV can recur in many women even after successful treatment. In addition, antibiotics can destroy healthy bacteria and therefore may eliminate the microbial defense system that healthy bacteria provide. This means that women with BV have an increased risk for contracting sexually transmitted infections, such as chlamydia, gonorrhea, and HIV. Pregnant women with BV are also at higher risk of preterm birth and other obstetric complications.
In order to reestablish healthy vaginal flora, an NIAID-supported research company called Osel has developed a novel product called LACTIN-V. LACTIN-V consists of a selected strain of Lactobacillus crispatus, one of the bacteria that protect the vagina from invading pathogens, that has been freeze-dried and formulated as a powder. When inserted into the vagina using a specially designed applicator, LACTIN-V helps reestablish the population of these beneficial bacteria. In early studies, LACTIN-V was shown to be safe and well-tolerated. Women who used LACTIN-V and became vaginally colonized with Lactobacillus crispatus had a lower rate of BV reinfection than those who didn't. LACTIN-V has also been shown to reduce the recurrence of urinary tract infections (UTIs) in women with a history of UTIs.
NIAID funded a Phase 2b clinical trial that showed that women who used LACTIN-V after a course of antibiotic treatment had a significantly lower risk of BV reinfection for at least 12 weeks. A substudy of this clinical trial found that LACITN-V also reduced two biomarkers associated with increased risk of contracting HIV following BV infection, suggesting that LACTIN-V might help prevent HIV infection in people with BV. In a non-NIAID clinical trial sponsored by Imperial College London, LACTIN-V reduced the rate of preterm birth among high-risk women in the Phase 1 trial. Further clinical trials are currently underway and are helping to build toward FDA approval. If successful development continues, this new product could be a significant advance in BV treatment and in women's health overall.


